- Urinary Incontinence and voiding dysfunction
- Men: Stress Urinary Incontinence
- Men: Pelvic Floor Rehabilitation
- Men: Transurethral Injection of Bulking Agents
- Men: Male Slings
- Men: Artificial Urinary Sphincter (AUS)
- Men: Adjustable continence device (Pro-ACT ) (Investigational)
- Women: Stress Urinary Incontinence
- Women: Pelvic Floor Rehabilitation
- Women: Transurethral Injection of Bulking Agents
- Women: Minimally Invasive Slings
- Women: Adjustable Continence Device
- Urge Incontinence in men and women
- Behavior Modification, Pelvic floor Rehabilitation and Biofeedback
- Botox Injection
- Sacral Neuromodulation
- Percutenous Tibial Nerve Simulation (PTNS)
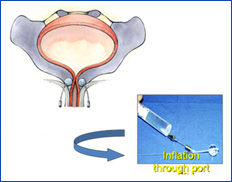
Women: Adjustable continence device (ACT ) (Investigational)
ACT or adjustable continence therapy is a new and innovative device that is currently under FDA investigation. Dr. Aboseif is one of the primary investigators evaluating this device and he is the primary author of the leading article looking at One Year Results of the North America ACT® Clinical Study Group: Aboseif, S.R; Franke, E.: The Adjustable Continence Therapy System: One Year Results of the North America Clinical Study Group.. J. Urol. May;181 (5):2187-91., 2009. The device consists of 2 silicon balloons filled with normal saline, placed percutenously at the level of the bladder neck. They will provide support both to the bladder neck and the urethra. The tension inside the balloon can be adjusted in the office to provide the optimum tension to adequately control the incontinence.